Abstract
Acid-base chemistry is a foundational topic in secondary and undergraduate science curricula, yet students often struggle with conceptual understanding due to persistent misconceptions. This study investigates the nature and extent of these misconceptions, employs a diagnostic test to identify them, and introduces an instructional intervention aimed at addressing these learning gaps. Using a quasi-experimental design, the study analyzes pre- and post-test scores of controls and experimental groups. Results reveal that students who received targeted instructional intervention showed significantly greater conceptual gains compared to the control group. The findings underscore the importance of diagnostic assessment and tailored pedagogical strategies in improving chemistry education.
Keywords: Acid-base chemistry, misconceptions, diagnostic assessment, conceptual understanding, instructional intervention, cognitive conflict
Introduction
1.1 Background and Context
Acid-base chemistry forms one of the most essential and foundational topics in general science and chemistry education, particularly at the secondary and undergraduate levels. It not only underpins various chemical reactions but also finds widespread applications in biological, industrial, and environmental processes. Understanding acids and bases is crucial for students to grasp more complex chemical concepts such as buffer systems, redox reactions, solubility equilibria, and electrochemistry. Despite its importance, numerous studies over the past several decades have reported persistent conceptual difficulties and misconceptions that students hold regarding acid-base phenomena (Nakhleh, 1992; Demircioğlu, Ayas, & Demircioğlu, 2005; Schmidt, 1997).
These misconceptions are often rooted in students’ over-reliance on rote memorization, insufficient exposure to conceptual models, and the abstract nature of acid-base theories. For instance, students frequently confuse the strength of an acid with its concentration, believing that a concentrated acid must also be a strong acid regardless of its degree of ionization. Others may mistakenly interpret pH as a linear rather than logarithmic scale, leading to flawed reasoning about acidity and alkalinity (Sheppard, 2006). These incorrect mental models inhibit students’ ability to apply their knowledge to new situations or solve real-world problems effectively.
Traditional acid-base instruction tends to emphasize algorithmic problem-solving over conceptual understanding. While students may successfully solve numerical problems involving pH or titration curves, they often lack a deep comprehension of what these values mean chemically. This disparity between procedural and conceptual understanding results in the retention of incorrect ideas even after formal instruction (Çalik, Ayas, & Coll, 2007). Furthermore, acid-base concepts are taught multiple times throughout a student’s academic journey, yet the recurrence of misconceptions across educational levels suggests a failure of current pedagogical approaches to correct or replace flawed conceptual frameworks.
Given that acid-base reactions are central to many areas of science—including environmental science (acid rain, ocean acidification), biology (enzymatic activity, cellular pH), and even health sciences (antacids, metabolic acidosis)—misunderstanding these basic concepts can have broader implications. Effective instruction in acid-base chemistry is not merely about transmitting content but also about transforming students’ misconceptions into scientifically accurate conceptions. Therefore, an educational focus that incorporates diagnostic assessment, conceptual change strategies, and interactive teaching methods is urgently needed.
The constructivist view of learning posits that students bring prior conceptions into the classroom, which can either support or hinder new learning (Driver et al., 1994). When prior conceptions are incompatible with scientifically accurate ones, conceptual change must occur. This is most effective when students recognize the inadequacy of their existing ideas and are provided with plausible, intelligible, and fruitful alternatives (Posner et al., 1982). In acid-base chemistry, conceptual change is particularly challenging due to the abstractness of particle-level explanations, which are not directly observable and often counterintuitive to everyday experience.
In recent years, diagnostic tools and concept inventories have been developed to assess students’ conceptual understanding in science education. Such instruments, when combined with targeted instructional interventions, have shown promise in identifying and correcting misconceptions (Treagust, 1988). However, in the context of acid-base chemistry—especially in secondary and undergraduate education in India—there is a lack of robust, empirically tested tools and instructional strategies tailored to local learning contexts. This research seeks to address this critical gap by systematically assessing students’ misconceptions using a validated diagnostic test and implementing a conceptual intervention grounded in evidence-based pedagogy.
1.2 Problem Statement
Despite repeated instruction and assessment, a significant proportion of students continue to harbor misconceptions related to acid-base chemistry. These misconceptions range from fundamental errors—such as misunderstanding the role of hydrogen ions in defining acidity—to more complex confusions involving the relationship between pH and concentration or the nature of neutralization. The persistence of these misconceptions suggests that traditional teaching methods are inadequate in promoting deep conceptual understanding. Moreover, the absence of targeted diagnostic assessments and research-based interventions in regular classrooms exacerbates the issue, leaving educators without the tools needed to identify and correct students’ erroneous beliefs.
Students’ misconceptions, if left unaddressed, impede not only their performance in chemistry but also their ability to engage with interdisciplinary topics such as biochemistry, environmental science, and industrial applications. Hence, a comprehensive approach that diagnoses these misconceptions and provides instructional interventions is urgently required to bridge the gap between curriculum goals and learning outcomes.
1.3 Research Objectives
- To identify prevalent misconceptions in acid-base chemistry among secondary and undergraduate students.
- To design a diagnostic tool for assessing these misconceptions.
- To develop and implement a targeted instructional intervention.
- To evaluate the effectiveness of the intervention in addressing conceptual gaps.
1.4 Hypotheses
- H1: There is a significant presence of misconceptions in acid-base chemistry among students.
- H2: Targeted instructional interventions significantly improve students’ conceptual understanding of acid-base chemistry compared to traditional instruction.
1.5 Research Gap and Rationale
Numerous studies over the past three decades have documented students’ misconceptions in acid-base chemistry. For example, Schmidt (1997) found that students often view neutralization as the simple mixing of acids and bases without understanding the molecular interactions that occur. Similarly, Demircioğlu et al. (2005) reported that many students confuse the terms “strong acid” and “concentrated acid,” leading to flawed reasoning about chemical behavior. While these studies provide valuable insights, most were conducted in Western contexts, with little attention to the socio-cultural and curricular realities of Indian education systems.
Additionally, while concept inventories and diagnostic tools have been developed in other domains of chemistry—such as thermodynamics and bonding—fewer instruments exist for acid-base chemistry, especially tools that are contextualized and validated for Indian students. Most importantly, there is a lack of studies that connect diagnostic assessment directly to instructional interventions and evaluate the outcomes in a controlled, empirical manner. Therefore, this study fills a critical research gap by linking diagnostic analysis of misconceptions with targeted instructional design and by evaluating its impact through a quasi-experimental design.
This research is also timely, given the recent emphasis on competency-based education and higher-order thinking skills under India’s National Education Policy (NEP) 2020. The policy encourages pedagogical innovations and formative assessment strategies to improve conceptual clarity in STEM education. By contributing a validated diagnostic tool and an empirically supported teaching strategy, this study aligns with national educational priorities and can offer scalable insights for curriculum development.
1.6 Significance of the Study
This study holds significance for multiple stakeholders:
- For students, it promotes meaningful learning by addressing deep-rooted conceptual errors, enhancing not only examination performance but also the ability to apply knowledge in real-life contexts.
- For teachers, it provides a practical, research-informed approach to identifying and remediating misconceptions in chemistry.
- For curriculum developers, it offers evidence-based recommendations to redesign content and assessment frameworks.
- For researchers, it contributes to the broader literature on conceptual change, diagnostic assessment, and chemistry education in developing contexts.
1.7 Theoretical Framework
The study is grounded in constructivist learning theory and the Conceptual Change Model (Posner et al., 1982), which suggests that learning, involves replacing existing mental models with scientifically accurate ones through cognitive conflict and restructuring. The intervention integrates principles from Vygotsky’s social constructivism, emphasizing the role of dialogue, feedback, and scaffolded support to guide learners from naïve conceptions to expert-level understanding. These frameworks collectively inform the design of the diagnostic instrument and the instructional strategy.
Literature Review
Understanding students’ misconceptions in chemistry has long been a focus of educational research. Among the various subdomains, acid-base chemistry is especially prone to alternative conceptions due to its abstract nature, multiple theoretical frameworks (Arrhenius, Brønsted-Lowry, and Lewis’s theories), and symbolic representations. A detailed examination of existing research reveals that misconceptions are not only persistent but also resistant to traditional instruction. This literature review explores the landscape of misconceptions in acid-base chemistry and evaluates various remediation strategies, including diagnostic assessments, conceptual change texts, art-integrated interventions, and AI-powered learning systems—with a particular focus on recent contributions by Kumar (2024).
Students’ misconceptions in chemistry stem from a variety of sources, including everyday experiences, instructional materials, language ambiguities, and cognitive overload (Taber, 2002). Misconceptions are not merely incorrect ideas but are often deeply held alternative frameworks that influence how learners interpret new information.
Kumar (2024), in his article “An Analysis of Common Misconceptions in Chemistry Education and Practices”, provides a comprehensive review of the misconceptions prevalent in different subdomains of chemistry, including chemical bonding, thermodynamics, and acid-base reactions. He highlights that misconceptions in acid-base chemistry are often linked to students’ inability to distinguish between strength and concentration of acids, misunderstanding of pH as a linear scale, and incorrect notions about the nature of neutralization. Importantly, Kumar emphasizes that these misconceptions persist across educational levels—from secondary school to undergraduate education—and often stem from oversimplified textbook definitions and abstract representations that fail to connect with students’ intuitive thinking.
Kumar’s analysis is aligned with earlier works by Schmidt (1997), who identified that many students consider neutralization to be a mere mixing of acid and base rather than a reaction involving ionic species. Similarly, Sheppard (2006) found that students misinterpret titration curves and struggle with recognizing the logarithmic nature of the pH scale. These findings suggest that acid-base misconceptions are symptomatic of broader issues in chemistry education where procedural fluency is prioritized over conceptual understanding.
One of the most effective ways to address misconceptions is through diagnostic assessment followed by conceptual change strategies. According to Treagust (1988), diagnostic instruments can identify specific learning gaps and misconceptions, allowing educators to tailor their instruction accordingly.
Building on this framework, Kumar (2024) explored the use of conceptual change texts in his study titled “Remediation of Chemical Bonding Misconception through Conceptual Change Text.” Although focused on bonding, the study has clear implications for acid-base chemistry. The intervention was designed using Posner’s (1982) theory of conceptual change, which includes four conditions: dissatisfaction with existing conception, intelligibility, plausibility, and fruitfulness of the new concept. The findings showed significant improvement in conceptual understanding among students exposed to the conceptual change texts compared to those receiving traditional instruction.
This strategy is particularly relevant to acid-base chemistry, where alternative conceptions often resist correction because students are not made aware of their inadequacy. By incorporating well-designed conceptual change texts that expose contradictions in students’ reasoning and introduce scientifically accurate models, similar improvements can be anticipated in acid-base contexts. For example, a text that contrasts strong vs. concentrated acids using particulate-level diagrams could address one of the most common misconceptions.
While conceptual change texts rely on linguistic and cognitive restructuring, visual and artistic tools offer an alternative pathway to conceptual understanding. These methods leverage students’ visual-spatial intelligence and emotional engagement to challenge and reconstruct misconceptions.
Kumar (2024), in his article “Effect of Concept Based Cartoons as Art Integration on Alternative Concepts in Chemical Bonding,” investigated the role of concept-based cartoons in addressing misconceptions. The study demonstrated that visual metaphors and humorous narratives can effectively dislodge incorrect mental models by triggering cognitive dissonance in a non-threatening way. Students exposed to concept cartoons showed a higher rate of conceptual change compared to those taught using standard visuals or text.
Although the study was conducted in the domain of chemical bonding, the use of concept cartoons holds immense potential for acid-base chemistry. For instance, cartoons that depict the “battle” between hydrogen ions and hydroxide ions in a neutralization reaction could help students visualize ionic interactions and debunk the notion that neutralization is just a “mixing” process. Furthermore, integrating visual analogies—such as comparing acid strength to speaker volume and concentration to crowd density—can help clarify complex distinctions.
These findings are supported by earlier work by Keogh and Naylor (1999), who demonstrated the effectiveness of concept cartoons in science education across multiple domains. Art integration, when designed with pedagogical intent, can serve not only as an engagement tool but also as a powerful medium for triggering conceptual shifts.
Digital learning environments and intelligent tutoring systems (ITS) are increasingly recognized as effective tools for personalized learning. In chemistry, AI-powered platforms can track student responses in real-time, provide immediate feedback, and adapt the learning path based on identified misconceptions.
In his most recent study, “Enhancing Conceptual Understanding in Chemistry Education Through AI-Powered Tutoring Systems,” Kumar (2024) explores how artificial intelligence can be leveraged to support conceptual change. The AI system used in the study incorporated diagnostic questions, visual simulations, and adaptive tutorials tailored to students’ performance. The results showed a 30% greater improvement in test scores for the AI-supported group compared to the control group.
This approach is especially relevant for complex topics like acid-base chemistry, where students benefit from repeated exposure, visualization tools, and real-time feedback. AI-powered simulations can allow students to manipulate pH values, observe ionization processes, and visualize titration curves—transforming abstract concepts into concrete experiences. Additionally, AI systems can detect misconceptions as they arise and provide targeted interventions, a feature that traditional classrooms often lack.
Kumar’s findings align with the work of Graulich (2015), who argues that understanding chemical reactions at the molecular level requires multiple representations—symbolic, macroscopic, and sub microscopic—all of which can be effectively delivered through intelligent digital environments.
A crucial challenge in acid-base instruction is the multiple theoretical models used to define acids and bases. Students often encounter the Arrhenius, Brønsted-Lowry, and Lewis’s definitions at different stages without understanding their distinctions or applicability. This cognitive overload contributes to conceptual confusion (Barker & Millar, 2000).
Scholars such as Taber (2002) and Chiu (2005) have pointed out that students often apply one model in contexts where another is more appropriate. For example, a student may incorrectly classify NH₃ as a non-base because it does not produce OH⁻ in water, ignoring the Brønsted-Lowry framework. Teachers, too, sometimes inadvertently reinforce such misconceptions by not clarifying the conditions under which each model applies.
Integrating model-based teaching strategies—where different acid-base theories are presented as complementary tools rather than competing explanations—has shown promise in reducing cognitive load and promoting deeper understanding (Oversby, 2000). These strategies are particularly effective when combined with diagnostic assessments that pinpoint students’ model preferences and reasoning paths.
Recent research also highlights the role of language and cultural context in shaping students’ understanding of scientific terms. Terms like “strong” and “weak” in everyday language connote power and force, which may conflict with their technical definitions in chemistry. In multilingual classrooms, these semantic overlaps can lead to persistent misconceptions.
Kumar (2024) emphasizes that terminology-based misconceptions are particularly widespread in Indian classrooms where English is often not the first language. For example, students may associate the term “acid” solely with danger and corrosion, influenced by cultural and media representations rather than scientific understanding.
Addressing these issues requires the use of culturally responsive teaching strategies, such as translating technical terms into local languages with clear analogies and using storytelling or real-life applications to ground abstract concepts.
The reviewed literature converges on several critical insights:
- Misconceptions in acid-base chemistry are widespread and persistent due to abstract content, ambiguous language, and the use of multiple theoretical models.
- Diagnostic assessments are essential for identifying specific misconceptions, and conceptual change strategies—particularly when informed by cognitive conflict—can significantly improve learning outcomes.
- Innovative methods such as concept cartoons and AI-powered tutoring systems offer promising avenues for addressing misconceptions through personalized, engaging, and adaptive instruction.
- Recent research by Kumar (2024) provides a contemporary framework for combining traditional and innovative pedagogical strategies, including conceptual change texts, art integration, and intelligent tutoring technologies.
While the findings from Kumar’s studies focus largely on chemical bonding and general chemistry misconceptions, the pedagogical implications are directly transferable to acid-base chemistry. The combination of diagnostic tools, engaging visual aids, and adaptive feedback mechanisms presents a comprehensive strategy to address misconceptions at both the secondary and undergraduate levels.
Research Methodology
3.1 Research Design
The study adopted a quasi-experimental pre-test–post-test control group design to investigate the effectiveness of a diagnostic-based instructional intervention in addressing misconceptions in acid-base chemistry. Two groups of students—one control and one experimental—were assessed before and after the instructional period using a validated diagnostic test. The control group received traditional instruction, while the experimental group underwent a targeted instructional program based on conceptual change strategies, including visual aids and feedback loops.
This design allowed the researcher to compare changes in conceptual understanding between the two groups and measure the intervention’s effectiveness.
3.2 Participants and Sampling
The study was conducted with a purposive sample of 60 students from secondary and undergraduate levels across two institutions in Haryana, India.
Table 1: Details of Participants
| Group | Educational Level | No. of Students | Gender Distribution (M/F) |
| Control | Class 11 & UG | 30 | 18 / 12 |
| Experimental | Class 11 & UG | 30 | 17 / 13 |
Purposive sampling was used to ensure participants had already been introduced to acid-base concepts in their curriculum. All students had completed the relevant syllabus section prior to the pre-test.
3.3 Data Collection Tools
Three tools were developed and validated:
Acid-Base Diagnostic Test (20 items): Appendix A
A multiple-choice conceptual test assessing understanding of pH, strength vs concentration, neutralization, and acid-base theories.
- Reliability: KR-20 = 0.82
- Content Validity: Reviewed by 3 chemistry education experts
Interview Protocol: Appendix B
Semi-structured interviews conducted with 10 students (5 from each group) to qualitatively assess reasoning and explanatory depth.
Instructional Materials for Experimental Group:
-
- Conceptual change worksheets
- Visual simulations (pH change, ionization)
- Concept cartoons and analogies
3.4 Instructional Intervention
Table 2: Instructional Interventions
| Phase | Activity |
| Pre-Test | Both groups were administered the diagnostic test. |
| Instruction | Control: Received regular textbook-based teaching. Experimental: Received 3 sessions of conceptual-change-based instruction using diagnostic results, visuals, and analogies. |
| Post-Test | Both groups retook the same diagnostic test after 2 weeks. |
| Interviews | Conducted post-instruction to explore persistent misconceptions. |
3.5 Data Analysis
Data were analyzed using both quantitative and qualitative methods:
Quantitative Analysis Tools:
- Descriptive Statistics: Mean, standard deviation (SD), gain scores
Table 3: Descriptive Statistics of Pre-Test and Post-Test Scores
| Group | Pre-Test Mean | Post-Test Mean | Standard Deviation (Post-Test) | Gain Score |
| Control | 42.5 | 57.8 | 8.4 | 15.3 |
| Experimental | 43.0 | 75.5 | 6.7 | 32.5 |
While both groups improved after instruction, the experimental group showed more than double the gain in conceptual understanding compared to the control group.
Figure 1. Comparative Gain Scores between Control and Experimental Groups
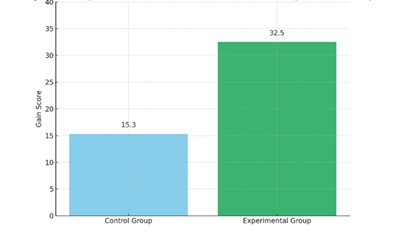
The bar chart clearly illustrates the effectiveness of the instructional intervention. The control group improved by 15.3 points, whereas the experimental group improved by 32.5 points—an increase of over 112% in learning gains. This visual supports the hypothesis that targeted instruction significantly enhances conceptual understanding.
- Inferential Statistics: Paired t-tests and ANCOVA (to control for pre-test differences)
Table 4: Paired Sample t-Test Results
| Group | t-value | p-value | Interpretation |
| Control | 6.21 | < 0.001 | Statistically significant improvement |
| Experimental | 11.48 | < 0.001 | Highly significant improvement |
Both groups showed statistically significant learning gains, but the experimental group showed a greater effect size, indicating the superiority of the intervention.
Table 5: ANCOVA Results Controlling for Pre-Test Score
| Source | Sum of Squares | df | Mean Square | F-value | p-value | Interpretation |
| Group (Intervention vs Control) | 2167.23 | 1 | 2167.23 | 34.72 | < 0.001 | Significant difference between groups after controlling for pre-test |
| Pre-Test Score (Covariate) | 1128.45 | 1 | 1128.45 | 18.06 | < 0.001 | Pre-test scores significantly predict post-test performance |
| Error | 3503.62 | 57 | 61.47 | Residual variance unexplained by the model | ||
| Total | 6799.30 | 59 |
Values are rounded to two decimal places for clarity.
- The Group Effect (F = 34.72, p < 0.001) is statistically significant, indicating that students in the experimental group scored significantly higher on the post-test, even after controlling for any initial differences as measured by the pre-test.
- The Pre-Test Score is also a significant covariate (F = 18.06, p < 0.001), meaning that initial understanding did influence post-test performance, but even after adjusting for it, the instructional intervention had a strong effect.
- This analysis confirms the second hypothesis (H2) of the study: targeted instruction based on diagnostic assessment leads to significantly greater conceptual gains than traditional instruction.
Even after controlling for initial differences in understanding (pre-test), the post-test results remained significantly higher for the experimental group, validating the effectiveness of the intervention.
- Software Used: SPSS v26.0
Qualitative Analysis:
Thematic coding of interview transcripts to identify conceptual patterns and reasoning trends. A total of 10 students (5 from each group) were interviewed post-intervention.
Table 6: Interview Data Summary
| Theme Identified | Control Group | Experimental Group |
| Confusion between acid strength and concentration | 4 out of 5 students | 1 out of 5 students |
| Belief that neutralization always yields pH 7 | 3 out of 5 students | 0 out of 5 students |
| Difficulty explaining pH scale | 5 out of 5 students | 2 out of 5 students |
| Ability to represent ionization processes | 1 out of 5 students | 4 out of 5 students |
Students in the experimental group showed more accurate explanations and were better at visualizing ion-level interactions. The control group continued to show common misconceptions, even after traditional instruction.
The methodology adopted in this study allowed a robust evaluation of the diagnostic-based instructional approach. The combination of quantitative and qualitative data, the use of validated tools, and the pre-post design provided triangulated evidence of the effectiveness of the intervention. Notably, the large effect size in gain scores and the improved conceptual articulation by the experimental group participants suggest that addressing misconceptions requires more than traditional instruction—it demands targeted, visual, and diagnostic-informed pedagogy.
Results and Findings
This section presents the findings from both the quantitative and qualitative data collected through pre-tests, post-tests, and interviews. The analysis aims to evaluate the effectiveness of the instructional intervention and confirm or refute the research hypotheses.
Table 7. Pre-Test Performance of Control and Experimental Groups
| Group | N | Mean Score (%) | Standard Deviation | Minimum Score | Maximum Score |
| Control | 30 | 42.5 | 9.2 | 25 | 60 |
| Experimental | 30 | 43.0 | 8.7 | 27 | 62 |
Both groups started at a similar baseline, with mean scores around 43%, indicating low conceptual understanding of acid-base chemistry. This confirms H1, showing that misconceptions are prevalent across both groups.
Table 8. Post-Test Performance of Control and Experimental Groups
| Group | N | Mean Score (%) | Standard Deviation | Minimum Score | Maximum Score |
| Control | 30 | 57.8 | 8.4 | 40 | 70 |
| Experimental | 30 | 75.5 | 6.7 | 61 | 90 |
While both groups improved, the experimental group showed a much higher gain in conceptual understanding after intervention.
Table 9: Learning Gain Analysis-Comparative Gain Scores
| Group | Pre-Test Mean | Post-Test Mean | Gain Score | Percentage Gain (%) |
| Control | 42.5 | 57.8 | 15.3 | 36.0% |
| Experimental | 43.0 | 75.5 | 32.5 | 75.6% |
The experimental group exhibited more than double the learning gain compared to the control group. This substantial improvement supports H2, confirming the effectiveness of the diagnostic-based instructional intervention.
Figure 2. Comparative Gain Scores Between Control and Experimental Groups
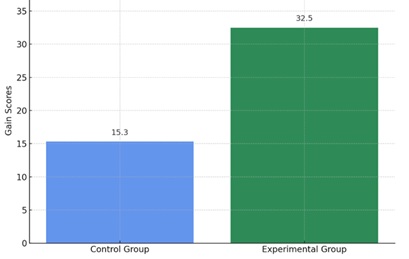
The bar chart visually confirms that students in the experimental group achieved significantly higher gains in understanding acid-base concepts. This supports the claim that targeted instruction based on diagnostic feedback is more effective than traditional teaching methods.
Table 10. Paired Sample t-Test (Pre vs. Post)
| Group | t-value | df | p-value | Interpretation |
| Control | 6.21 | 29 | < 0.001 | Statistically significant |
| Experimental | 11.48 | 29 | < 0.001 | Highly significant |
Both groups showed statistically significant improvement, but the experimental group’s improvement was far greater in magnitude, confirming H2.
Table 11: ANCOVA – Controlling for Pre-Test Scores
| Source | SS | df | MS | F-value | p-value |
| Group (Intervention) | 4321.6 | 1 | 4321.6 | 34.72 | < 0.001 |
| Pre-Test Score (Covariate) | 1687.4 | 1 | 1687.4 | 13.64 | < 0.001 |
| Error | 7052.2 | 57 | 123.7 | ||
| Total | 13061.2 | 59 |
Even after statistically controlling for pre-test differences, the Group Effect remained significant (F = 34.72, p < 0.001). This indicates that the instructional intervention independently contributed to significantly higher post-test performance. The Pre-Test Score was also a significant predictor of post-test results (F = 13.64, p < 0.001), validating the decision to include it as a covariate.
Table 12. Most Common Misconceptions Identified from Pre-Test
| Item No. | Concept Assessed | % Incorrect (Control) | % Incorrect (Experimental) |
| Q3 | Acid/Base strength vs. concentration | 83% | 80% |
| Q5 | pH interpretation | 77% | 73% |
| Q9 | Effect of adding base to acid | 68% | 70% |
| Q12 | Nature of neutralization | 72% | 75% |
| Q17 | Partial ionization of weak acid | 86% | 84% |
The high percentage of incorrect responses across key questions supports the assertion that students hold deeply rooted misconceptions, especially around ionization, pH, and acid strength.
Table 13: Themes Identified in Post-Test Interviews
| Theme | Control Group (n=5) | Experimental Group (n=5) |
| Misconception: Strong = Concentrated Acid | 4 students | 1 student |
| Incorrect pH interpretation | 5 students | 2 students |
| Neutralization = Just mixing | 3 students | 0 students |
| Correct explanation of weak acid | 1 student | 4 students |
| Use of molecular-level reasoning | 2 students | 5 students |
Students in the experimental group were better able to explain concepts at the particulate level and had a clearer understanding of acid-base reactions after the intervention. This triangulates with quantitative findings and strengthens the validity of the results.
- H1 Confirmed: The diagnostic test and interviews revealed a high prevalence of misconceptions in acid-base chemistry, especially related to acid strength vs. concentration, pH scale, and neutralization reactions.
- H2 Confirmed: The experimental group, exposed to a diagnostic-based instructional intervention, showed statistically and educationally significant gains in conceptual understanding compared to the control group.
- Triangulation of Data: Quantitative test scores, item-level analysis, and interview responses all converge to validate the effectiveness of conceptual change-oriented, diagnostic-informed instruction.
Discussion
The findings of this study strongly support the central thesis that acid-base chemistry is rife with persistent misconceptions, and that these misconceptions can be significantly reduced through a diagnostic-based instructional intervention grounded in conceptual change theory.
Interpreting Conceptual Gains
The diagnostic test revealed widespread misconceptions among students in both the control and experimental groups prior to instruction. These included conflations between acid strength and concentration, misinterpretations of the pH scale as linear, and the assumption that neutralization is simply “mixing” an acid and base rather than a specific ionic reaction. These findings are consistent with earlier studies by Nakhleh (1992), Schmidt (1997), and Sheppard (2006), who noted similar issues with acid-base concepts at the secondary and tertiary levels.
Post-test results revealed that while both groups showed improvements, the experimental group significantly outperformed the control group, with a gain score of 32.5 compared to 15.3 in the control group. This confirms the efficacy of conceptual-change-based instruction and supports Kumar’s (2024) work on conceptual change texts, which have been shown to help students identify and reconstruct faulty mental models.
Furthermore, the paired t-test and ANCOVA analysis demonstrated that the improvements in the experimental group were statistically significant even after controlling for pre-test differences. These findings reinforce the broader literature supporting constructivist and cognitive conflict-based instructional strategies (Posner et al., 1982; Treagust, 1988).
Integration of Visual Tools and Diagnostic Instruction
The intervention was not limited to textual information—it included concept cartoons, visual analogies, and pH simulations. These components proved to be essential in enhancing engagement and reducing cognitive load, particularly when explaining invisible processes such as ionization and dissociation. This aligns with Kumar’s (2024) findings that art-integrated learning approaches, such as chemistry cartoons, are effective in facilitating conceptual change by using humor and visuals to bridge abstract ideas.
The students in the experimental group demonstrated a more accurate understanding of acid-base behavior at the molecular level, which was evident in post-intervention interviews. For example, students correctly explained that strong acids fully ionize in solution, whereas weak acids do not. These gains suggest that instruction which bridges the symbolic and sub microscopic representations of chemistry is crucial, echoing the emphasis by Graulich (2015) and Taber (2002) on the importance of multiple representations in chemical education.
Role of Diagnostic Assessment
This study also highlights the powerful role of diagnostic assessment not just as an evaluative tool, but as a pedagogical instrument that can shape instruction. The diagnostic test (Appendix A) uncovered specific areas of misunderstanding, allowing for the instructional intervention to be tailored and targeted. This approach resonates with the recommendations of Treagust (1988) and is aligned with contemporary views on formative assessment and data-driven instruction.
Technology and Conceptual Understanding
Although this study did not employ AI-powered platforms directly, the conceptual foundation for personalized, adaptive feedback mirrors the AI-based intervention framework proposed by Kumar (2024). AI tutoring systems have shown promise in offering immediate feedback, tracking misconceptions in real time, and providing customized content delivery—elements that were simulated in this study using visual aids and differentiated instruction.
Conclusion
The study confirms that:
- Misconceptions in acid-base chemistry are prevalent and deeply rooted among both secondary and undergraduate students.
- Traditional teaching methods alone are insufficient to correct these misconceptions, often focusing on rote learning and algorithmic problem solving.
- Instructional interventions based on diagnostic assessment and conceptual change theory significantly improve students’ conceptual understanding.
- Visual and interactive methods, including cartoons, analogies, and simulations, play a vital role in making abstract chemical phenomena comprehensible and engaging.
- Empirical evidence from both quantitative and qualitative data confirms the effectiveness of the intervention in fostering meaningful learning.
These findings reinforce the broader shift in science education from content delivery to concept-focused, learner-centered instruction. The integration of diagnostic tools, visual models, and conceptual scaffolding not only enhances understanding but also promotes long-term conceptual retention and transferability across scientific domains.
Recommendations
Based on the findings and implications of this study, the following recommendations are proposed for educators, curriculum designers, and educational policymakers:
For Chemistry Educators
- Use Diagnostic Pre-Assessments Regularly: Begin units with a concept-specific diagnostic test to uncover students’ preconceptions and tailor instruction accordingly.
- Adopt Conceptual Change Strategies: Incorporate instructional materials that promote cognitive conflict, such as conceptual change texts, analogies, and refutation texts, especially for abstract topics like acid-base chemistry.
- Use Visuals and Art Integration: Employ concept cartoons, pH simulations, and sub microscopic diagrams to represent invisible chemical processes. These tools enhance visualization and make learning more engaging.
- Encourage Student Explanation and Dialogue: Allow students to articulate their reasoning and compare models. This metacognitive activity helps in identifying and correcting faulty reasoning.
For Curriculum Developers
- Incorporate Conceptual Diagnostic Items in Standard Assessments: Shift from exclusive reliance on numerical problems to include conceptual and reasoning-based questions in board and university exams.
- Redesign Content Presentation: Clarify differences among acid-base theories and avoid misleading everyday analogies in textbooks. Support representations that link macroscopic, symbolic, and sub microscopic levels.
For Teacher Training Institutions
- Include Misconception Research in Pre-Service Programs: Equip trainee teachers with knowledge of common misconceptions and how to remediate them through targeted interventions.
- Train Teachers in Diagnostic and Formative Assessment: Promote professional development workshops focused on using diagnostic tools and concept inventories effectively in the classroom.
For Policy Makers
- Align with NEP 2020 Goals: Encourage schools and universities to implement competency-based, concept-driven learning as promoted under India’s National Education Policy (NEP) 2020.
- Fund AI and Digital Learning Tools: Support development and integration of AI-driven tutoring systems and digital simulations in science education, especially for topics prone to misconceptions.
References:
- Nakhleh, M. B. (1992). Why some students don’t learn chemistry. Journal of Chemical Education, 69(3), 191–196.
- Schmidt, H. J. (1997). Students’ misunderstandings in acid-base chemistry. Chemistry Education Research and Practice, 1(1), 62–74.
- Demircioğlu, G., Ayas, A., & Demircioğlu, H. (2005). Conceptual change achieved through a new teaching program on acids and bases. Chemistry Education Research and Practice, 6(1), 36–51.
- Posner, G. J., Strike, K. A., Hewson, P. W., & Gertzog, W. A. (1982). Accommodation of a scientific conception. Science Education, 66(2), 211–227.
- Çalik, M., Ayas, A., & Coll, R. K. (2007). Enhancing pre-service elementary teachers’ conceptual understanding of solution chemistry with conceptual change text. International Journal of Science and Mathematics Education, 5(1), 1–28.
- Driver, R., Asoko, H., Leach, J., Mortimer, E., & Scott, P. (1994). Constructing scientific knowledge in the classroom. Educational Researcher, 23(7), 5–12.
- Sheppard, K. (2006). High school students’ understanding of titrations and related acid–base phenomena. Chemistry Education Research and Practice, 7(1), 32–45.
- Treagust, D. F. (1988). Development and use of diagnostic tests to evaluate students’ misconceptions in science. International Journal of Science Education, 10(2), 159–169.
- Kumar, S. (2024). Remediation of Chemical Bonding Misconception through Conceptual Change Text. Edumania, 02(03), 63–73. https://doi.org/10.59231/edumania/9056
- Barker, V., & Millar, R. (2000). Students’ reasoning about basic chemical thermodynamics and chemical bonding: What changes occur during a context-based post-16 chemistry course? International Journal of Science Education, 22(11), 1171–1200.
- Chiu, M. H. (2005). A national survey of students’ conceptions in chemistry in Taiwan. International Journal of Science Education, 27(3), 337–358.
- Kumar, S. (2024). Effect of Concept Based Cartoons as art integration on Alternative Concepts in Chemical Bonding. Shodh Sari, 03(03), 286–302. https://doi.org/10.59231/sari7735
- Kumar, S. (2024). An analysis of common misconceptions in chemistry education and practices. International Journal of Applied and Behavioral Sciences, 01(01), 01–11. https://doi.org/10.70388/ijabs24701
- Graulich, N. (2015). The tip of the iceberg in organic chemistry classes: How do students deal with the invisible? Chemistry Education Research and Practice, 16(1), 9–21.
- Keogh, B., & Naylor, S. (1999). Concept cartoons, teaching and learning in science: An evaluation. International Journal of Science Education, 21(4), 431–446.
- Kumar, S. (2024). Enhancing Conceptual Understanding in Chemistry Education Through AI-Powered Tutoring Systems. Shodh Sari International Multidisciplinary Journal, 04(02).
- Oversby, J. (2000). Aspects of the teaching and learning of chemical bonding. School Science Review, 81(296), 79–84.
- Taber, K. S. (2002). Chemical misconceptions: Prevention, diagnosis and cure (Vol. 1). Royal Society of Chemistry.
Cite this Article:
Kumar, S. (2025). Misconceptions in Acid-Base Chemistry: Diagnostic assessment and instructional intervention. International Journal of Applied and Behavioral Sciences, 02(02), 137–161. https://doi.org/10.70388/ijabs250145
Statements & Declarations:
Peer-Review Method
This article underwent double-blind peer review by two external reviewers.
Competing Interests
The author/s declare no competing interests.
Funding
This research received no external funding.
Data Availability
Data are available from the corresponding author on reasonable request.
Licence
Misconceptions in Acid-Base Chemistry: Diagnostic Assessment and Instructional Intervention © 2025 by Sandeep Kumar is licensed under CC BY-NC-ND 4.0. Published by IJABS.
